Why Eradicate BVD?
Of the many infectious diseases that affect cattle, BVD is once again in the spotlight. Whilst various eradication schemes have been in place across the UK for a number of years now there is still a drive to increase engagement with these projects. In England the long-awaited Animal Health and Welfare Pathway (AHWP) provides funding towards veterinary input and diagnostics as part of the Sustainable Farming Incentive. For the infectious diseases element of the AHWP in cattle the focus is on BVD. A yearly vet visit valued at £372 for dairy and £522 for beef producers is available to all herds with 10 or more cows that are currently eligible for the BPS. This review is carried out by the farms’ own vet and will allow them, for the BVD element, to review biosecurity measures testing and create a bespoke control plan.
Why Eradicate BVD?
There are several reasons to choose to eradicate BVD:
- Firstly, it’s relatively easy to identify and remove persistently infected (PI) cattle, which are the main source of BVD infection.
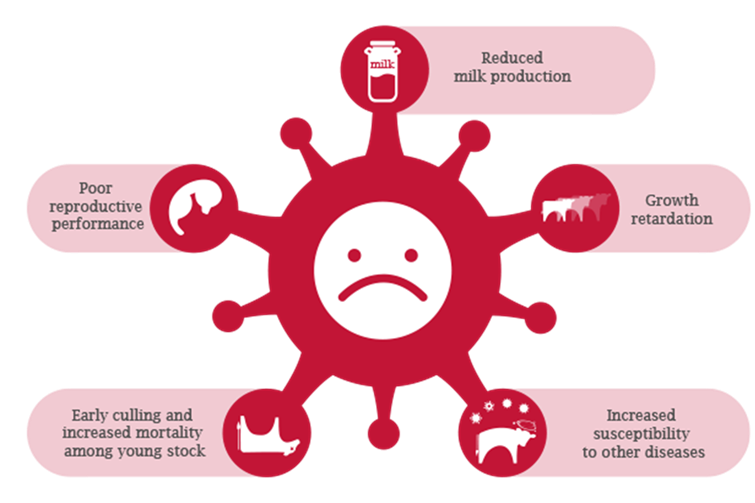
- Secondly, BVD suppresses the immune system making cattle more susceptible to other disease, so by eradicating BVD, other diseases become easier to control.
- Thirdly, BVD has a serious financial impact. Research has shown a 5-fold return on investment of eradicating BVD from suckler herds, and a 14-fold return for dairy herds1; much of this is due to improvements in herd fertility and calf health.
There are wide ranging economic impacts of BVD on a herd, whether dairy or beef. It is difficult to accurately estimate all these costs - some are direct and measurable such as abortion, milk loss and infertility, however, others are very difficult to measure, like immunosuppressive effects and poorer milk quality. An analysis of 31 papers looking at the economic impact of BVD gave a mean cost of £46.50 per cow per year2, but figures varied widely depending on, for example, whether disease was endemic within herds or if it was a new outbreak in a naïve herd. In a recent Europe-wide study carried out in BVD endemically affected dairy herds, it was shown that vaccinated animals within these herds produced up to 1.8L milk/day more during early lactation than their unvaccinated counterparts in the same herds, even in the absence of clinical signs of BVD3.
Risk Factors
Neighbouring herds, buying in stock, sending pregnant heifers to graze elsewhere, shared equipment and people can all pose a BVD infection risk. The biosecurity review will determine how BVD could potentially enter your herd and what can be done to mitigate these risks.
Testing
Testing is required to establish the BVD status of your herd and to identify any PI cattle within the herd. The removal of PI animals is absolutely essential and the first step to becoming BVD free. Some tests such as bulk milk testing in dairy herds or youngstock blood tests are paid for as part of the review in the AHWP - speak to your vet for advice on the best testing regime for your herd.
If all testing is negative that’s great news, but, it’s really important not to stop here. All herds regardless of status should have a robust plan to keep BVD out by tightening up biosecurity, continuing to monitor and using vaccination as an insurance policy.
Biosecurity
Having a farm specific biosecurity plan is key to keeping BVD out. This will be tailored to suit the biosecurity risks that are identified for your herd alongside general biosecurity measures that will help to protect the health of your cattle. The aim of a biosecurity plan is not to expect perfect biosecurity – it’s to be aware of where risks exist and make plans to mitigate those risks. By plugging these gaps new BVD outbreaks in naïve herds and breakdowns in herds where BVD has been eradicated become much less likely.
Vaccination
For most herds, vaccination is an important part of their BVD control strategy. It is vital that all breeding cows and heifers are fully protected throughout pregnancy to prevent the birth of PI calves. Bovela® is a gene-deleted live vaccine against BVD which prevents the birth of PI calves caused by transplacental infection (given at least 3 weeks prior to service). Bovela® is administered in a single injection, so there are no concerns regarding timings of multiple doses in the primary course, and an annual single dose booster ensures this protection continues. The ease of use means less handling of animals which is beneficial to cattle and humans alike! Vaccination can be employed in herds where disease already exists to prevent the birth of further PIs and perpetuation of the virus whilst these animals are identified and removed, and in herds that have eradicated or are naïve to BVD to ensure they remain free and don’t suffer the often hugely damaging impact of a new BVD outbreak.
The AHWP review is available now in England, providing funding to help you improve your herd health status. BVD is a hugely economically impactful disease, but it is a disease that can be controlled by testing to establish status, reviewing biosecurity practices, vaccination and creating a robust control plan that is specific your herd and farm. Contact your vet for further information on how to protect your herd.
References
- Stott et al. (2012 ) Predicted costs and benefits of eradicating BVDV from Ireland I Vet J, 65:12
- Yarnall & Thrusfield (2017) Engaging veterinarians and farmers in eradicating bovine viral diarrhoea: a systematic review of economic impact Vet Rec;181(13):347
- Schmitt-van de Leemput et al. (2020) Comparison of milk production of dairy cows vaccinated with a live double deleted BVDV vaccine and non-vaccinated dairy cows cohabiting in commercial herds endemically infected with BVD virus PLoS ONE 15(10): e0240113.
Bovela® contains modified live BVDV*-1, non-cytopathic parent strain KE-9:104.0-106.0 TCID50**, Modified live BVDV*-2, non-cytopathic parent strain NY-93:104.0-106.0 TCID50**. * Bovine viral diarrhoea virus **Tissue culture infectious dose 50%. UK: POM-V. Advice should be sought from the prescriber. Further information available in the SPC or from Boehringer Ingelheim Animal Health UK Ltd., RG12 8YS, UK. Tel: 01344 746957. Email: vetenquiries@boehringer-ingelheim.com. Bovela® is a registered trademark of Boehringer Ingelheim Vetmedica GmbH, used under licence ©2023 Boehringer Ingelheim Animal Health UK Ltd. All rights reserved. Date of preparation: June 2023. BOV-0082-2023. Use Medicines Responsibly.
- 2nd October 2023

